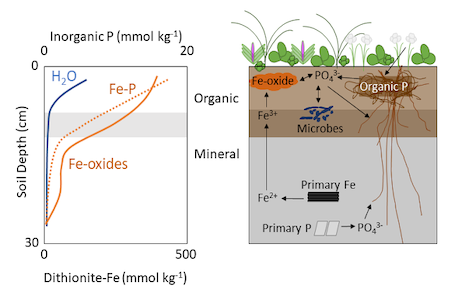
NGEE Arctic
Next-Generation Ecosystem Experiments
Advancing the predictive power of Earth system models through understanding
of the structure and function of Arctic terrestrial ecosystems

Advancing the predictive power of Earth system models through understanding
of the structure and function of Arctic terrestrial ecosystems